Dallas is a large city in the state of Texas, USA. It is most famous for its oil and cotton industries and its busy railroad lines. The city has population of around 1,300,000 people and is home to a large amount of Fortune 500 companies. In Dallas, there are 337 public schools, almost 90 private schools and 38 colleges. Dallas is the centre of education in the southern central United States. The University of Texas Southwestern Medical School is an exclusive medical school admitting only about 200 students per year. Other universities include Texas Women’s University which is a nursing school located in Parkland Memorial Hospital, and Dallas Baptist University which is a private university that offers Biblical and Christian subjects. It enrols around 5,500 people and provides students with graduate, doctoral and undergraduate degrees. Criswell College is a university started by the First Baptist Church of Dallas in the 1970s, also offering Biblical studies and has around 400 students enrolled currently. Dallas Theological Seminary offers students seminaries in the evangelical faith and boasts an alumni of around 12,000 people. There are a number of universities in the close proximity of Dallas including The University of Dallas in Irving and the El Centro College. El Centro College was established in 1966 and has a student population of around 10,000 students and as is one of the only universities in Dallas which offers fashion design and marketing programs as well as food hospitality programs. The main school district of Dallas is the Dallas Independent School District which enrols over 160,000 students. The School for the Talented and Gifted in Dallas was voted the best school in the United States numerous times and another Dallas school, the Science and Engineering Magnet has been named number two in the very same list on numerous occasions. There are a high number of private schools in Dallas, including The Hockaday School, Burton Adventist Academy, Parish Episcopal School, The Winston School, Yavneh Academy of Dallas and Dallas Lutheran School.
Free Wi-Fi
To make sure you’re always connected we offer completely free and easy to access wi-fi.








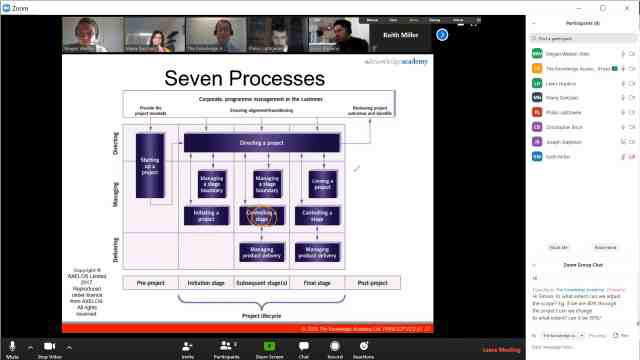
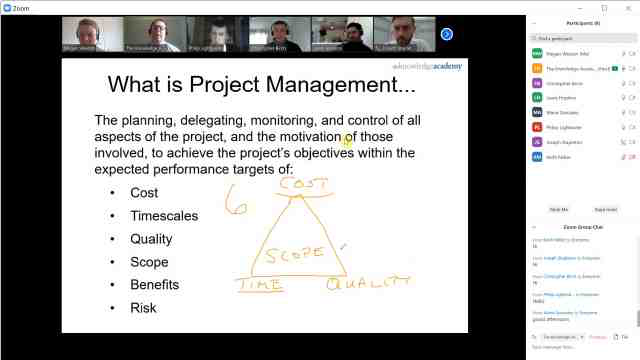
























 Back to course information
Back to course information




 If you wish to make any changes to your course, please
If you wish to make any changes to your course, please

